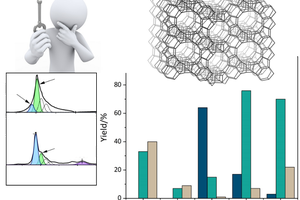
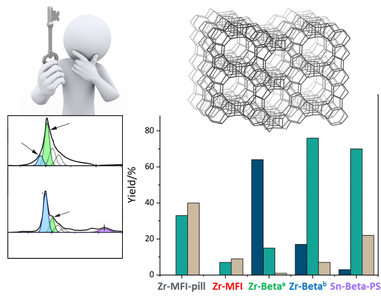
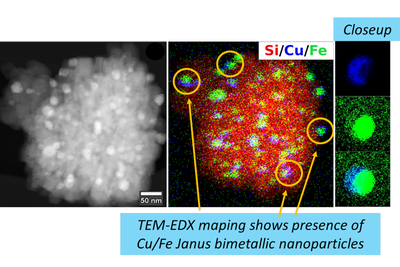
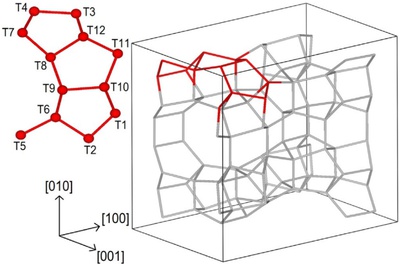
Zeolites are porous elementosilicates with vast applications in the fields of catalysis, sorption, and ion-exchange. Their advantage lies in properties, such as high adsorption capacity, defined microporosity (resulting in molecular sieving effect), adjustable chemical composition, and possibility for post-synthesis modifications. Conventional synthesis of zeolites uses solvothermal methods that are limited in term of control and design of the process. On the contrary, recently developed ADOR synthesis approach (Assembly, Disassembly, Organization, Reassembly) gives the possibility to design the final products and prepare different type of porous materials by post-synthesis manipulations (using 2D zeolite precursors) or removal of specific building units from the parent material. Design and synthesis of novel zeolitic materials and their advanced characterization by electron microscopy and electron diffraction methods are general aims of proposed PhD thesis. The synthesized materials will be investigated for their potential use e.g. in catalysis.
The PhD work will include:
- the application of established synthetic techniques (e.g. solvothermal synthesis, ADOR approach, swelling, pillaring, and functionalization,
- development of novel synthesis strategies, and
- characterization of synthesized material by electron microscopy and diffraction methods (including novel techniques like Continuous Rotation Electron Diffraction (cRED).
The PhD work will result in preparation of new zeolitic materials and their structure determination by advanced characterization methods.
The experience in electron microscopy and zeolite synthesis will be an advantage for the potential candidate.