What is the mechanism by which proteins cyclin Y and 14-3-3 activate the protein kinase CDK16?
We are proud to share the news that a new research article of Prof. Tomáš Obšil from our Department and of and Dr. Veronika Obšilová from the Institute of Physiology of the CAS has been published in prestigious journal Nature Communications.
Abstract
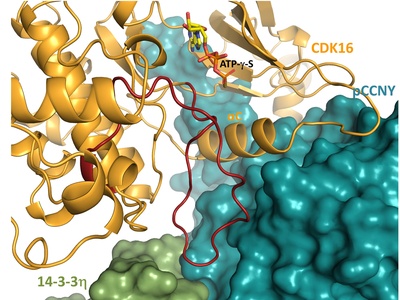
Cyclin-dependent protein kinase 16 (CDK16) regulates both physiological and pathological processes, including autophagy, spermatogenesis and cancer. Unlike other CDKs, CDK16 is regulated by an unclear mechanism involving phosphorylated cyclin Y (pCCNY) in complex with 14-3-3 proteins rather than CCNY alone. In this study, research teams from the Department of Physical and Macromolecular Chemistry (website) and the Institute of Physiology of the CAS (website) have elucidated this mechanism by structurally and functionally characterizing CDK16 in complex with pCCNY and 14-3-3. Utilizing cryo-EM analysis and hydrogen/deuterium exchange coupled to mass spectrometry, the researchers demonstrated that 14-3-3 binding modulates the conformation of a key moiety of the CDK binding surface of pCCNY, thereby enabling CDK16 activation. These findings not only clarify the role of pCCNY and 14-3-3 in CDK16 activation but also highlight the potential of targeting CDK16 protein-protein interactions for cancer therapy.